 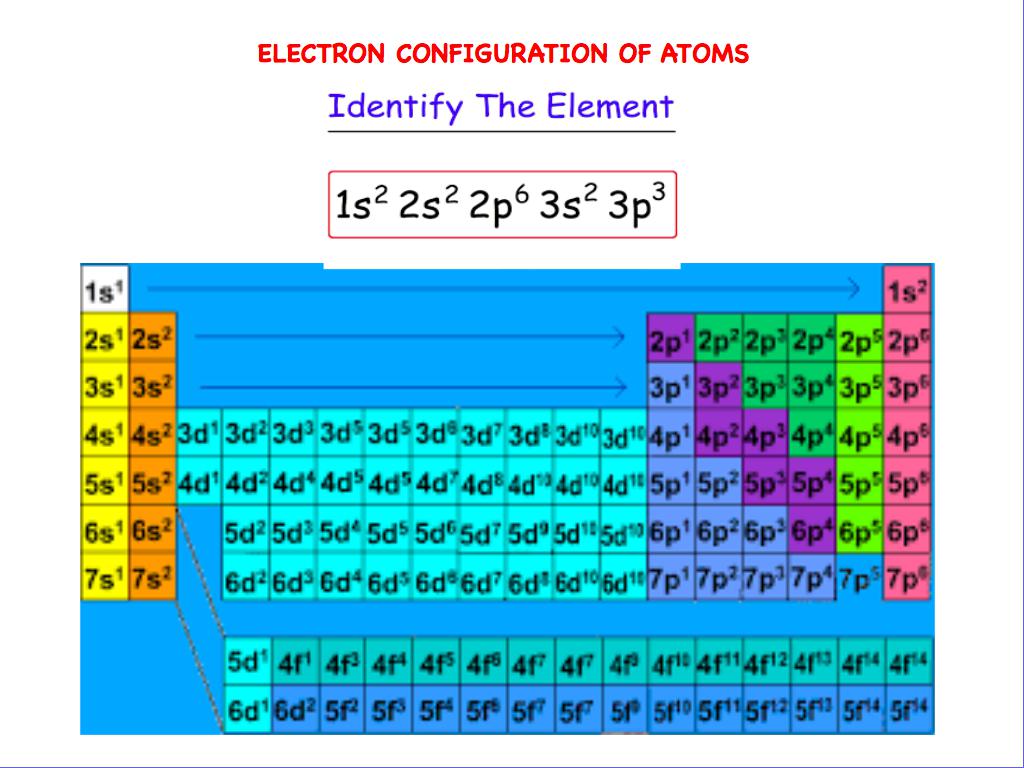
The orbital labels s, p, d, and f originate from a now-discredited system of categorizing spectral lines as sharp, principal, diffuse, and fundamental, based on their observed fine structure. Thus, the electron configuration of neon is pulled out, and phosphorus is written as follows: 3 s 2 3 p 3.Īn even simpler version is simply to quote the number of electrons in each shell, e.g. Phosphorus, for instance, differs from neon (1 s 2 2 s 2 2 p 6) only by the presence of a third shell. It is often abbreviated by noting that the first few subshells are identical to those of one or another noble gas. Phosphorus (atomic number 15), is as follows: 1 s 2 2 s 2 2 p 6 3 s 2 3 p 3.įor atoms with many electrons, this notation can become lengthy. Lithium has two electrons in the 1s subshell and one in the (higher-energy) 2 s subshell, so its ground-state configuration is written 1 s 2 2 s 1. An atom's subshells are written in order of increasing energy – in other words, the sequence in which they are filled (see Aufbau principle below).įor instance, ground-state hydrogen has one electron in the s orbital of the first shell, so its configuration is written 1s 1. In this notation, a subshell is written in the form nx y, where n is the shell number, x is the subshell label and y is the number of electrons in the subshell. Physicists and chemists use a standard notation to describe atomic electron configurations. This information can be written as 5s 2 5p 6 5d 10 5f 14 5g 18 (see below for more details on notation). Here is the electron configuration for a filled fifth shell: If the states also share the same value of m, they are said to lie in the same atomic orbital.īecause electrons have only two possible spin states, an atomic orbital cannot contain more than two electrons ( Pauli exclusion principle).Ī subshell can contain up to 4 l+2 electrons a shell can contain up to 2 n² electrons where n equals the shell number. States with the same value of n and also l are said to lie within the same electron subshell, and those electrons having the same n and l are called equivalent electrons. States with the same value of n are related, and said to lie within the same electron shell. In large atoms, shells above the second shell overlap (see Aufbau principle). Shells and subshells (also called energy levels and sublevels) are defined by the quantum numbers, not by the distance of its electrons from the nucleus, or even their overall energy. No two electrons in one atom can have the same set of these four quantum numbers ( Pauli exclusion principle). s and l in part determine the electron's magnetic dipole moment. 
Spin is an intrinsic property of the electron and independent of the other numbers. +½ or -½ (sometimes called "up" and "down") (s=0, p=1.)ĭetermines energy shift of an atomic orbital due to external magnetic field ( Zeeman effect). The orbital's angular momentum, also seen as the number of nodes in the density plot. Partly the overall energy of the orbital, and by extension its general distance from the nucleus. Three of these are integers and are properties of the atomic orbital in which it sits (a more thorough explanation is given in that article). The state of an electron in an atom is given by four quantum numbers. The discussion below presumes knowledge of material contained at Atomic orbital. 1.5 Relation to the structure of the periodic table.In bulk materials this same idea helps explain the peculiar properties of lasers and semiconductors. The concept is also useful for describing the chemical bonds that hold atoms together. Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. Because of the Pauli exclusion principle, no more than two electrons may exist in a given atomic orbital therefore an electron may only leap to another orbital if there is a vacancy there. The probability that the act of measurement will detect the electron at a particular point in space is proportional to the square of the absolute value of the wavefunction at that point.Įlectrons are able to move from one energy level to another by emission or absorption of a quantum of energy, in the form of a photon. According to the Copenhagen interpretation of quantum mechanics, the position of a particular electron is not well defined until an act of measurement causes it to be detected. 
Formally, the quantum state of a particular electron is defined by its wavefunction, a complex-valued function of space and time. Like other elementary particles, the electron is subject to the laws of quantum mechanics, and exhibits both particle-like and wave-like nature. In atomic physics and quantum chemistry, the electron configuration is the arrangement of electrons in an atom, molecule, or other physical structure ( e.g., a crystal). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
